Boilers for burning solid fuels and combustion in them
Most solid fuel boilers can operate on several types of fuel - brown coal, anthracite, coke, wood, fuel briquettes, peat, etc. (Fig. 16). The calorie content of different types of fuel differs significantly, so the boiler power and operating time from one load on different types of fuel will be different.
Rice. 16. Various types of solid fuel for boilers
When purchasing a boiler, a buyer often thinks that the boiler’s power will be the same, regardless of the type of fuel burned. In reality, the drop in power when burning less calorie fuel can reach 25-30% (subject to the permissible fuel moisture content). When the boiler operates on fuel with a humidity of 30-40%, the loss of power will be even greater.
Manufacturers indicate which fuel must be used as the main fuel, and which fuel can be used as an additional fuel. It is advisable to use the main type of fuel; the boiler can operate on it for a long time without harm to it.
The range of solid fuel boilers is represented by two types of boilers: made of cast iron or steel. There is no clear answer as to which material is better suited for a solid fuel boiler (cast iron or steel).
Manufacturers specialize in both cast iron and steel boilers. When choosing a solid fuel boiler, first of all, it is necessary to take into account the main characteristics and tasks that the boiler must cope with: power, type of fuel, loading chamber volume, combustion duration, efficiency and safety, as well as price. Understanding the material characteristics of a solid fuel boiler is necessary for its proper operation and maintenance.
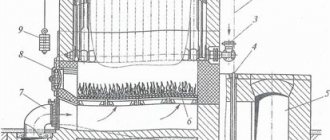
The cast iron boiler is a sectional structure. Each section can be separated from each other. The dismountable design is more convenient when transporting, installing or servicing the boiler in case of its breakdown or depressurization (Fig. 17).
Rice. 17. Design and main elements of a cast iron boiler
During operation, cast iron becomes covered with dry rust, which is a film of iron oxide (this is the so-called chemical corrosion). As a rule, dry rust does not progress.
Cast iron undergoes wet corrosion much more slowly than steel. Cast iron heat exchangers can be cleaned less frequently - their efficiency, even due to the appearance of carbon deposits during operation, decreases less.
A minor disadvantage of a cast iron heat exchanger is the possibility of thermal shock: if cold water enters a cast iron heat exchanger that has not cooled down, it may crack, so large temperature differences between the supply and return lines should be avoided. The thermal inertia of cast iron boilers is higher; they take a long time to heat up, but on the other hand, they cool down more slowly.
The steel boiler is a one-piece monoblock, which is assembled and welded in a factory.
Boilers made of steel are less susceptible to destruction from sudden temperature changes. Steel is more elastic than cast iron and can easily withstand temperature differences, even if the boiler is fed with cold water into the return line. The ability of steel to easily withstand temperature changes allows solid fuel boilers with a steel heat exchanger to more widely use boiler automation. Steel boilers react faster than cast iron boilers, they cool faster and heat up faster.
However, we should not forget that with large and frequent temperature changes, “fatigue zones” may appear in steel and, as a result, cracks in places weakened by welding. If a steel boiler is rusted or damaged by deposits of hardness salts, then repair will most likely be simply impossible - getting factory-quality welding at home is almost impossible. In fact, a burnt-out steel boiler will have to be thrown away, whereas in a cast-iron boiler the repairman will simply replace the damaged section.
Traditional solid fuel boilers (Fig. 18) in their design resemble a conventional stove with a window for fuel supply, a firebox and a chimney. The fuel for a traditional solid fuel boiler can be either coal or wood. The main element of a traditional solid fuel boiler is a heat exchanger, which ensures the transfer of thermal energy to the coolant.
The obvious advantage of a traditional low-power solid fuel boiler, suitable for individual heat supply, is the absence of electronic boards, automation and all kinds of control systems, which are the first to fail. The only automation device is a temperature controller that operates on a mechanical principle, so a traditional solid fuel boiler is not only quite versatile, but also reliable, capable of operating for a long time without maintenance.
Rice. 18. Traditional solid fuel boiler design
Pyrolysis boilers produce thermal energy by burning solid fuel. A pyrolysis boiler has a higher efficiency, and, therefore, more thermal energy can be obtained from a smaller volume of wood than with the same operation of a traditional boiler.
Wood combustion occurs in three phases according to the principle of gas formation when burning wood:
1 - drying firewood;
2 - degassing (approximately 85% of substances during the combustion of wood turn into flammable gases, the rest remain charcoal);
3 - combustion - at temperatures above 600 °C, combustible gases oxidize and ignite and a burning layer of charcoal is formed. From approximately 900 oC to 1000 oC, thermal decomposition of charcoal and oxidation of carbon in it occurs.
The fan in the boiler directs the flame downward, making the entire fuel combustion process controllable. In addition, a constant supply of oxygen to the combustion zone guarantees complete oxidation of combustible gases. For this purpose, in addition to the main air, preheated secondary air is supplied for post-combustion.
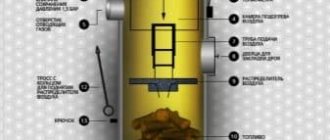
Long-burning boilers (top-burning boilers) are a type of solid fuel boiler in which the air supply and combustion process are limited to the upper part of the fuel layer. This scheme allows you to simultaneously load a significant amount of fuel into the firebox. Such boilers are characterized as long-burning boilers and require less frequent maintenance.
A pellet boiler is a solid fuel boiler that uses wood pellets as fuel. Wood pellets are made from wood waste: shavings, sawdust and other residues from the woodworking industry.
At its core, a pellet boiler is not much different from a traditional solid fuel boiler in which wood is burned. The main difference is that the pellet boiler, in addition to the combustion chamber, is equipped with a special hopper and automatic fuel supply (Fig. 19-20).
Rice. 19. Boiler with fuel bunker and automatic fuel supply
Rice. 20. Boilers with a fuel bunker and automatic fuel supply
Rice. 21. Boilers with a fuel bunker and automatic fuel supply
Fig. 22. Boiler with a fuel bunker and automatic fuel supply
Rice. 23. Diagram of the boiler and the processes occurring in it
Rice. 24. Construction of a boiler unit for burning wood
Rice. 25. Construction of a boiler for burning wood
Rice. 26. Construction of a boiler for burning wood
Rice. 27. Construction of a boiler for burning solid fuel
Figure 28. Construction of boilers for burning solid fuels
Rice. 29. Construction of boilers for burning solid fuels
Rice. 30. Construction of boilers for burning solid fuels
Rice. 31. Construction of boilers for burning wood
Rice. 32. Construction of a boiler for burning wood
Rice. 33. Solid fuel industrial boiler
Rice. 34. Boilers for working on different types of fuel (solid, liquid, gas)
Rice. 35. Construction of a boiler for burning solid fuel
Rice. 36. Construction of a boiler for burning wood (three types of boiler)
Rice. 37. Construction of long-burning wood-burning boilers
Rice. 38. Construction of a long-burning wood-burning boiler (two types)
Rice. 39. Construction of a long-burning wood-burning boiler (two types)
Fuel combustion process
Combustion of fuel is the process of chemical combination of its combustible elements with oxygen in the air, releasing heat and forming a flame. Fuel with a high yield of volatile substances and a low ignition temperature ignites faster, so firewood or other types of fuel with a low ignition temperature are used to ignite coal and anthracite in the firebox. Firewood under the influence of a torch of fire (heat source) quickly ignites and continues to burn with the release of more and more new portions of volatile substances, which, when ignited, increase the temperature in the fuel layer and, in turn, cause the release of volatile substances in the embedded portion of coal or anthracite, continuing combustion process.
The process of fuel combustion in the furnace proceeds as follows. Initially, the fuel is warmed up due to the heat received directly from both the hot lower layer of burning fuel and from the hot flue gases passing through the layer of fresh fuel; As the fuel warms up, it dries, i.e., moisture evaporates from it. Then the release of volatile combustible substances begins, which ignite and burn, usually above the layer. A non-volatile residue remains in the layer, consisting of coke (carbon) and ash. The coke burns in the layer, and the ash partially falls into the ash pan, from where it is removed, and partially it melts, turning into slag, which is removed from the grate during partial or complete cleaning of the firebox. The hot, burning layer of fuel, as well as its small particles in the firebox and volatile substances, burning in the combustion chamber, give off heat to the heating surfaces of the solid fuel boiler. As the fuel burns out, the remaining ash covers the fuel particles from oxidation by air and the fuel combustion rate decreases. When scuffed, pieces of fuel are freed from ash, again exposed to atmospheric oxygen, and combustion improves. The more ash there is in the fuel, the more often it has to be removed and the firebox cleaned.
To maximize the use of heat during fuel combustion, the following conditions are necessary:
- the dimensions of the firebox must correspond to the performance of the solid fuel boiler and the properties of the fuel;
- an even layer of fuel on the grate and its same composition in size, ensuring a uniform flow of air over the entire area of the grate;
- supply of the required amount of air to the firebox;
- the presence of high temperature in the firebox for preheating the fuel to the ignition temperature, as well as for the combustion of flammable volatile substances and small particles of unburned coal carried by gases and air from the fuel layer;
- the volume of the combustion space must ensure complete combustion of volatile fuel substances and its carbon particles before the flue gases come into contact with the heating surfaces.
Go to boiler selection >>
Depending on the nature of the combustion process, flaming and flameless combustion of fuel is distinguished.
During flaming combustion of fuel (wood, oil shale, peat, brown coal, long-flame and gas coals), a larger amount of volatile substances is released, combustion usually occurs in the combustion chamber, the bed temperature for most coals does not reach the value at which mineral impurities melt, and Only ash and debris remain on the grate. The resistance of the ash layer is low, and there are no special obstacles to the passage of air.
With flameless combustion, the fuel usually burns in a layer on the grate, producing only small tongues of light transparent flame (lean coals and anthracites). The temperature in the layer increases greatly, heating the grates and mineral impurities, which can melt and form slag. The slag layer and the layer of non-caking coke create great resistance to air passage. If there is insufficient amount of air entering the firebox, the temperature of the lower part of the layer increases, heating the grate even more.
To achieve the required performance of the heating boiler, it is sometimes necessary to increase the area of the grate to the maximum possible size (when burning brown coals and shale with loose slag and loose ash). Lengthening the grates can only be done if slag is removed from the furnace through the tilting or retractable part of the grate. If slag is removed through the front loading door of the furnace into the boiler room, then lengthening the grate complicates maintenance and does not bring significant benefit. To facilitate maintenance of the extended grate, you can install additional scoop doors on the side walls of the firebox and use them to remove slag from the end of the grate when cleaning the firebox. An increase in the area of the grate can also be achieved by increasing its width.
The draft in solid fuel boilers is created by a chimney or smoke exhausters. Draft in the chimney occurs because the heated flue gases are lighter than air and therefore they are forced into the chimney by the pressure of the heavier outside air. The higher the temperature of the flue gases and the higher the chimney, the stronger the draft and the faster the air enters the boiler furnace. When the chimney is not heated, the draft worsens; as it warms up, the draft increases.
When passing through the grate and the fuel layer, the air must overcome significant resistance. If there is a low chimney or a thick layer of fine fuel, the draft may be insufficient and an insufficient amount of air will enter the firebox, resulting in incomplete combustion of the fuel.
The draft is regulated by a damper (gate) on the chimney behind the boiler, the control of which is moved to the front of the boiler for convenient maintenance. Opening the smoke damper (damper) too large creates an increased vacuum in the firebox and causes an increased flow of cold air through leaks in the firebox, hatches, etc., which causes an increase in fuel consumption. Insufficient opening of the damper reduces the vacuum in the furnace and leads to incomplete combustion of the fuel, smoking and the ejection of flame through the combustion door into the boiler room. Leaks in the combustion door, solid fuel boiler lining, chimney, etc. impair draft due to air suction.
To overcome the high resistance of the fuel layer in the firebox with air, artificial blast is used with a fan supplying air under the grate (Figure 1) or the draft is increased by installing a smoke exhauster (Figure 2), which sucks in exhaust gases from the firebox and flues of the solid fuel boiler, directing them into the chimney.
The normal combustion process is achieved by regulating the air supply to the firebox using dampers on the chimney (damper) and on the air duct (throttle valve), while combustion should occur at the lowest possible vacuum in the firebox above the fuel layer. The completeness of fuel combustion can be judged by the color of the flame in the firebox and the color of the smoke coming out of the chimney.
A sign of complete combustion of the fuel is a light yellow flame in the firebox.
Signs of incomplete combustion of fuel in the firebox are:
- reddish flame with dark stripes, resulting from a lack of air and too thick a layer of fuel or slagging of the grate;
- blue flames or black smoke from the chimney, resulting from a lack of air caused by excessive slagging of the grate, too thick a layer of fuel when the boiler firebox is strongly forced, low temperature in the firebox;
- a bright white flame that occurs due to a large excess of air with a high boost of the firebox of a solid fuel boiler;
- light spots (fuel burns), through which a lot of air breaks through, cooling the firebox, while small pieces (fractions) of fuel are carried away by the air from the fuel layer.
Each flame color corresponds to a certain temperature in the firebox of a solid fuel boiler:
| Red color | 525° |
| Dark red color | 700° |
| Cherry color | 900° |
| Light cherry color | 1000° |
| Light yellow (light straw) color | 1200° |
| White color | 1300° |
| Bright white color | 1400° |
For complete combustion of fuel, it is necessary to correctly select the thickness of its layer, since small and large coals create unequal resistance to the passage of air for normal combustion. For fine fuel, you need to keep the layer on the grate thinner so that enough air can pass through the fuel layer. For large fuels, the layer must be kept thicker so that excess air does not pass through it, as this will lead to a decrease in temperature.
The amount of air required for complete combustion of 1 kg of fuel is called the theoretically required amount of air. However, complete combustion of the fuel cannot be achieved in this case, since it is practically difficult to achieve complete mixing of air with the fuel, so more air has to be supplied in order for complete combustion to occur.
The ratio of the actual amount of air entering the firebox to the theoretically required amount is called the excess air coefficient. The excess air coefficient for various fireboxes and fuels ranges from 1.3 to 1.6, i.e. 30–60% more air is supplied to the firebox than the amount it needs.
A minimum amount of excess air must be supplied to the firebox, since excess air cools the firebox and causes excessive fuel consumption. The air supply to the firebox is regulated by changing the draft and blast.
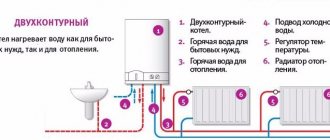
Boilers for burning liquid fuels and combustion in them
Liquid fuel boilers are structurally somewhat different from solid fuel boilers.
For universal boilers designed to burn liquid fuel, a special burner with a safety circuit breaker is supplied, without which the operation of the boilers is not allowed.
Combination boilers for heating a house can use not one, but several types of fuel. For this feature they are also called universal. Their main advantage is the ability to work during interruptions in the main type of fuel.
Depending on the fuel combinations, there are several types of boilers, which differ in design and type of burner.
A feature of liquid-fuel heating boilers is the use of fuel oil, diesel fuel or kerosene as fuel. The main chemical elements that make up any liquid fuel for heating boilers are oxygen, hydrogen, carbon and sulfur. Due to the presence of non-combustible minerals and moisture in fuel oil, ash is formed during the combustion of liquid fuel.
The ash content of fuel oils is mainly determined by the content of oxygen-containing compounds containing metal cations. Some ash comes from suspended particles (mainly silicates and silica). When moving to more viscous fuel oils, the content of suspended particles and colloidal particles increases. Ash is an extremely undesirable component of fuel oil combustion products, as it clogs nozzles, accelerates corrosion of equipment and requires periodic shutdown and cleaning of boiler equipment. The most dangerous are vanadium compounds (in ash they are represented by vanadium pentoxide V2O5), which sharply reduce the resistance of most steels to high-temperature corrosion.
The main properties of boiler oil as a liquid fuel, which determine its use in boiler plants, are: sulfur content, viscosity, pour point, ash content, lower calorific value, density.
Fuel oil is a mixture of hydrocarbons (with a molecular weight of 400 to 1000), petroleum resins (with a molecular weight of 500-3000 or more), asphaltenes, carbenes, carboids and organic compounds containing metals (V, Ni, Fe, Mg, Na, Ca). The physicochemical properties of fuel oil depend on the chemical composition of the source oil and the degree of distillate fraction distillation and are characterized by the following data: viscosity 8-80 mm2/s (at 100 °C), density 0.89-1 g/cm3 (at 20 °C) , pour point 10-40 °C, sulfur content 0.5-3.5%, ash up to 0.3%, lower calorific value 39.4-40.7 MJ/mol.
Based on the amount of sulfur, fuel oils are divided into low-sulfur, sulfur and high-sulfur with a sulfur content of up to 0.5%, 2% and 3.5%, respectively. The sulfur content in fuel oil depends on the source oil, but is significantly higher than in it, since sulfur is concentrated mainly in heavy residual products. When processing high-sulfur oils, the sulfur content in fuel oil can reach up to 4.3%. Sulfur is contained in fuel oils in active and passive forms. Active sulfur causes corrosion of pipelines, tank heaters, heat exchangers and tail heating surfaces at metal wall temperatures below the dew point temperature.
Fuel oils from oil refineries usually contain only traces of water. Fuel watering occurs during transportation and especially when heated with open steam. When heating fuel oil with open steam, the moisture content in the fuel oil increases sharply, which not only causes losses of steam and condensate, but also reduces the quality of the fuel oil itself. As a result, the efficiency of the boiler units and the reliability of the boiler room operation decreases. When heated in open tanks, the water contained in the fuel oil causes foaming.
Boiler fuel oil can be low-viscosity or high-viscosity with a high content of resinous substances and paraffin. The viscosity of fuel oil is an important operational factor that determines the ability to transport, drain, pump and burn it. With increasing temperature, the viscosity of fuel oil decreases, therefore all operations with fuel oil are carried out with heating.
Depending on the viscosity, boiler fuel oil comes in several grades, differing in their pour point, which is always above 0 °C. For the most viscous grades of fuel oil, the pour point is 25 °C and higher, so preheating of such fuel oil is necessary: when pumping up to 60-70 °C, and when burning up to 140 °C.
The flash point of fuel oil is the temperature at which its vapor forms a mixture with the surrounding air that ignites when a fire is brought to it.
When heating fuel oil in open (without pressure) containers, for fire safety purposes, the heating temperature should be approximately 10 °C below the flash point. In closed containers (coils, pipes) under pressure, fuel can be heated to fuel oil significantly above its flash point.
Currently, gas-oil burners of various designs are installed on water-tube boilers (DE, DKVR) and water heating units (KV-GM), which meet the requirements of economical and safe operation. The main thing is to ensure approximately equal combustion quality and flame length for both types of fuel (natural gas and fuel oil). Gas-oil burners are a complex of a gas burner and an oil nozzle and, depending on the design, are designed for separate or joint combustion of gas and liquid fuels. To install the burner in the front wall (lining) of the boiler, an embrasure is made.
In decentralized heat supply, as a rule, diesel fuel and light grades of fuel oil are used. First of all, this is due to the convenience of their transportation and storage, low viscosity, which facilitates the task of efficient combustion, as well as low sulfur and ash content, which solves the problem of environmental pollution and equipment safety.
Diesel boilers are boilers that run on diesel fuel, and it is always possible to replace a diesel burner with a gas burner (thanks to this, these boilers can be considered universal). Burners for diesel boilers are selected based on the power of the boiler. Diesel boilers are the optimal solution when gas has not yet been supplied, but it is planned to be supplied. In this case, you will not need to buy another boiler or change the heating and hot water supply system, but simply change the burner to a gas one.
Rice. 40. Construction of a boiler for burning liquid fuel
Rice. 41. Construction of boilers for burning liquid fuel
Figure 42. Construction of boilers for burning liquid fuel
Rice. 43. Construction of a boiler for burning liquid fuel (two types)
Rice. 44. Construction of boilers for burning liquid fuel
Regulating the amount of fuel burned
The combustion time and the amount of fuel burned in the furnace is regulated by the amount of air supplied to it. For solid fuel, for example, by increasing the air blow under the grate and draft, you can thereby introduce more air into the firebox. Air oxygen, passing through the fuel layer and reacting, is distributed into a large number of streams and increases the combustion rate. By throwing more fuel into the firebox, we get more heat. However, the required thickness of the fuel layer should be kept within established limits in order to avoid incomplete combustion (increased mechanical and chemical underburning of the fuel) or a large excess of air. The draft is adjusted so that in the firebox (in the upper part of the loading door) a vacuum of no more than 20-30 Pa (2-3 mm of water column) is established.
The combustion of gaseous fuel includes the following stages: mixing gas with air (formation of a combustible mixture), heating the mixture to the ignition temperature and combustion itself. Combustion can be complete or incomplete. During complete combustion of fuel, carbon combines with oxygen to form carbon dioxide (carbon dioxide) CO2, hydrogen - water vapor H2O, and sulfur - sulfur dioxide (sulfur dioxide) SO2. In this case, the gases leaving the furnace will not contain combustible elements. If the firebox is not properly maintained, incomplete combustion of fuel occurs when the chemical energy of the fuel is not completely converted into heat; and partly remains in the flue gases and is released through the chimney into the atmosphere.
Carbon monoxide, a product of incomplete combustion of fuel, is one of the air pollutants (loss from chemical incomplete combustion). Another atmospheric pollutant is small particles of unburned fuel carried along with gases (up to 80%) (losses from mechanical incomplete combustion, as well as carryover of ash and soot). The reason for the carryover of unburned fuel in low-power heating boilers is the combustion of ordinary fuels containing a large amount of fines.
Boilers for burning gas and combustion in them
Rice. 45. Construction of boilers for burning gaseous fuels
Rice. 46. Construction of boilers for burning gaseous fuels
Gaseous fuel
Gaseous fuel is a mixture of various gases: methane, ethylene and other hydrocarbons, carbon monoxide, carbon dioxide or carbon dioxide, nitrogen, hydrogen, hydrogen sulfide, oxygen and other gases, as well as water vapor.
Methane (CH4) is the main constituent of many natural gases. Its content in natural gases reaches 93...98%. When 1 m3 of methane is burned, ~35,800 kJ of heat is released.
Gaseous fuels may also contain small amounts of ethylene (C2H4). The combustion of 1 m3 of ethylene produces ~59,000 kJ of heat.
In addition to methane and ethylene, gaseous fuel also contains hydrocarbon compounds, for example propane (C3H8), butane (C4H10), etc. The combustion of these hydrocarbons releases more heat than the combustion of ethylene, but in combustible gases their amount is insignificant.
Hydrogen (H2) is 14.5 times lighter than air. When 1 m3 of hydrogen is burned, ~10,800 kJ of heat is released. Many flammable gases, except coke gas, contain relatively small amounts of hydrogen. In coke oven gas its content can reach 50...60%.
Carbon monoxide (CO) is the main flammable component of blast furnace gas. When 1 m3 of this gas is burned, ~12,770 kJ of heat is generated. This gas is colorless, odorless and very poisonous.
Hydrogen sulfide (H2S) is a heavy gas with an unpleasant odor and is highly toxic. The presence of hydrogen sulfide in the gas increases corrosion of the metal parts of the furnace and gas pipeline. The harmful effects of hydrogen sulfide are enhanced by the presence of oxygen and moisture in the gas. When 1 m3 of hydrogen sulfide is burned, ~23,400 kJ of heat is released.
The remaining gases: CO2, N2, O2 and water vapor are ballast components, since with an increase in the content of these gases in the fuel, the content of its combustible components decreases. Their presence leads to a decrease in the combustion temperature of the fuel. The content of free oxygen in gaseous fuel >0.5% is considered dangerous according to safety regulations.